What is Shared Clinical Decision Making?
Inside the MAHA movement's rebrand of informed consent.
Image credit: iStock.com/Fatcamera
I’ve asked a number of my colleagues if they could define “shared clinical decision-making,” and I couldn’t get two exact answers. Instead, I got responses like: Isn’t that what’s already happening with providers and patients? and You mean informed consent?
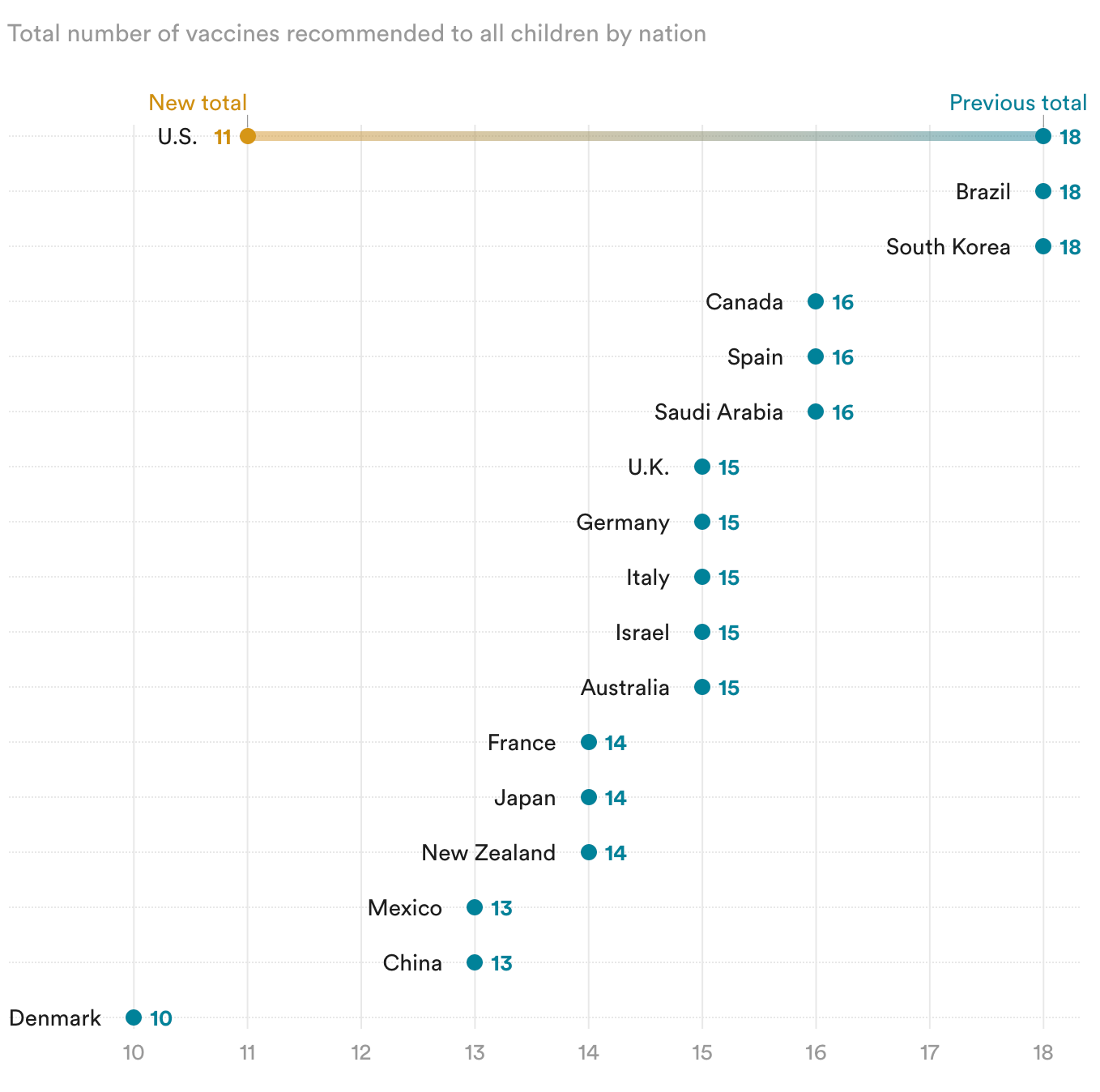
In January of 2026, the Centers for Disease Control and Prevention (CDC) slashed the number of recommended vaccines in the pediatric schedule from 17 to 11. It’s not lost on me that exactly one year earlier, in January of 2025, RFK Kr. said in his confirmation hearing, “I support the childhood schedule.” In that year, the science didn’t change—only the politics.
The decision to modify the schedule affected influenza, COVID-19, and RSV recommendations in the middle of respiratory virus season under the false narrative that we would be “aligning the U.S. childhood vaccine schedule with the international consensus while strengthening transparency and informed consent.”
Let’s be very clear: there is not a singular international vaccine schedule because every country tailors its vaccine schedule to the population’s health needs. On average, most countries vaccinate against 13 to 16 diseases. The smallest schedule we compared ours to was Denmark’s, where they vaccinate against 10 diseases. After being right in the middle of consensus, our pediatric vaccine schedule is now the outlier.
Image credit: STAT News
The decision also included a nod to a common trope in the anti-vaccine movement: that vaccines are given without proper informed consent.
Let’s be very clear again: Providers are required by federal law — under the National Childhood Vaccine Injury Act, NCIVA — to share Vaccine Information Statements (VIS) at every vaccination appointment. They can be shared in paper or digital format. VISs are information sheets produced by the CDC that explain, in detail, the risks and benefits of vaccines. If the person being vaccinated is under the age of 18, the VIS is given to the parent or caregiver.
So what about shared clinical decision-making?
In addition to massively reducing the vaccine schedule, the CDC said that parents should choose whether or not they want some of the previously recommended vaccines (like rotavirus, hepatitis A, hepatitis B, etc.) based on “shared clinical decision-making.” According to the CDC’s new webpage on this topic:
"Unlike route, catch-up, and risk-based recommendations, shared clinical decision making vaccinations are individually based and informed by a decision process between the health care provider and the patient or parent/guardian.” The webpage goes on to say that “ACIP makes shared clinical decision-making recommendations when individuals may benefit from vaccination, but broader vaccination of people in that group is unlikely to have population-level impacts.”
I must admit, my jaw nearly hit the floor when I read that. Especially, as it relates to the Hepatitis B vaccine, where we have years of data to show that early vaccination reduces the risk of chronic hepatitis B vaccine—at a population level—by 90%.
You might have read that and been confused. And to be honest, you wouldn’t be alone. Most patients/parents do not know what shared clinical decision-making really means. Instead of empowering them in their decision-making, it's actually causing more confusion. And we have some data on this.
In August and December of 2025, the Anneberg Public Policy Center (APPC) surveyed nearly 2,000 adults about their understanding of this new framing. The surveys showed significant gaps in understanding the specific context of whether to get a vaccine or not. Here are some of the findings:
1 in 5 people think shared clinical decision-making means that “taking the vaccine may not be a good idea for everyone, but would benefit some.”
More than 2 in 5 people think that shared clinical decision-making is “up to an individual whether to consult with their health care provider before taking a vaccine, about whether it would be a good idea.”
1 in 4 people think shared clinical decision making means they should discuss vaccination decisions with family members
1 in 10 people are unsure what shared clinical decision-making means for new vaccines
It’s exactly what we feared: it’s presenting the vaccine schedule as an à la carte menu.
When participants were asked about who should be a part of shared clinical decision-making, many were unsure. Most chose physicians, but only half included nurses, and only a third included pharmacists.
The introduction of shared clinical decision-making presents a false reality that there is no discussion happening between providers and patients. I can absolutely empathize with patients not feeling like they have enough time with their providers, but to claim that vaccinations are happening without informed consent and that this new framing will increase transparency and trust is both untrue and incredibly harmful.


Thank you for examining the nuance behind CDC’s new emphasis on this confusing phrase which is a “solution” creating new problems for public health policy.